NEW
Rapamycin Pilot Treatment Trial for ME/CFS
Hypothesis: Relief of symptoms via mTOR inhibition
The First Biomarker-Driven Treatment Trial for ME/CFS
Subsets Mean Success
Until now, the heterogeneity of the patient population and the lack of a reliable biomarker to diagnose and prognose ME/CFS has thwarted clinical trials for both non-approved FDA drugs as well as repurposed agents.
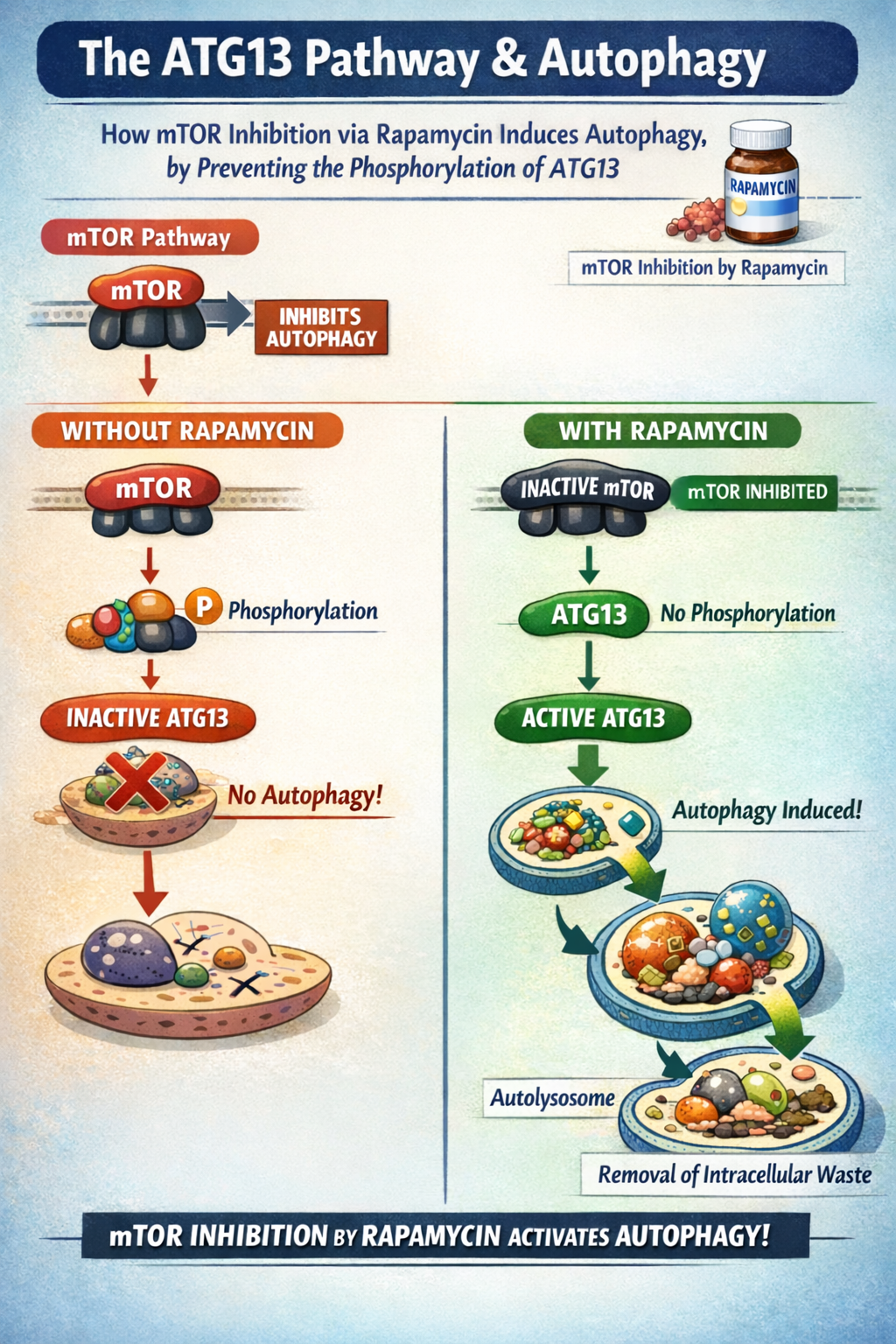
We identified elevated levels of inactive ATG-13 as a testable and targetable pathway for treating symptoms of post-exertional malaise.
Recently, our study published in Inflammation Research shows that lack of ATG13 inside cells induces macrophages to take on an inflammatory role, impairing myelin in muscles, in a process aggravated by exercise.
Biomarker to Treatment Trial
Rapamycin therapy inhibits mTOR and reduces autophagy disruption. We believe that a subset of patients may have chronic mTOR activation that can lead to the symptoms of ME/CFS. By taking rapamycin, the mTOR inhibitor, we hope that these people may see a significant reduction in symptoms.
We are tracking symptoms and autophagy markers in this study.
Why Rapamycin
Our publication on elevated ATG13 showed that a subset of ME/CFS patients display serological evidence of autophagy disruption. We have shown that this deficit in autophagy is due to the chronic activation of mTOR. Without properly functioning autophagy, there is significant cellular stress, immune activation, and not enough energy for the cell to do well.
Rapamycin is an mTOR inhibitor. It is an FDA approved drug that was initially developed to protect patients during a kidney transplant. It has a well understood safety profile. This study tracks autophagy markers and ME/CFS symptoms in patients who are treated with low-dose rapamycin by participating clinicians.
We published Phase I results in Journal of Translational Medicine showing some patients had improved symptoms of fatigue and PEM and that autophagy biomarkers correlated with improvement. Now we plan a double-blinded, placebo-controlled trial to test symptoms and biomarkers under rigorous conditions.
The Science: Simmaron Publications
Molecular and Cellular Neurosciences Elevated pATG13 in ME/CFS patients and microglial activation via RAGE receptor.
Immunologic Research Genetic depletion of ATG13 and/or mTOR-driven ATG13 dysfunction (pATG13) results in dysfunctional autophagy, resulting in the first in vivo evidence of PEM. Further, ATG13 dysfunction upregulates IL-6 and RANTES and leads to demyelinating pathology in the muscles serving nerves.
Inflammation Research Genetic depletion of the early autophagy protein ATG13 impairs mitochondrial energy metabolism, augments oxidative stress, induces the polarization of macrophages to the M1 inflammatory mode, and compromises myelin integrity in skeletal muscle.
Journal of Translational Medicine Phase I proof of concept observational pilot of low-dose rapamycin in ME/CFS and Long-COVID patients.
Our Goals
One goals are to demonstrate efficacy in a placebo-controlled trial, characterize a subset of patients who are likely to respond to this potential treatment, and develop a predictive test. These achievements would be much-needed firsts for ME and Long-Covid. We also intend to submit a grant for an NIH exploratory treatment trial based on our data. This is a big step in Simmaron’s overarching goal of spurring more treatment trials for ME/CFS patients who have waited far too long.
Participating Clinicians
David Kaufman MD, lead clinician Center for Complex Diseases (listen to his podcast)
Daniel Peterson MD Sierra Internal Medicine
Bela Chheda MD Center for Complex Diseases—Palo Alto, CA
Brayden Yellman MD Bateman Horne Center
Jennifer Bell APRN, FNB-BC Bateman Horne Center
Jon Berner MD, PhD Woodinville Psychiatric
Stephanie Grach MD, MS Mayo Clinic
Simmaron’s Role
Simmaron Research funds the low-dose rapamycin trial, as part of our non-profit mission to conduct scientific research that produces evidence-based treatments for ME/CFS and Long-Covid patients.
Our Research and Development lab at UWM is responsible for studying the blood and questionnaires. Our role is to collect and analyze data tracking symptoms and autophagy.
We hypothesize that ME/CFS patients with elevated serum levels of pATG-13 and related autophagy disruption markers who are given low dose rapamycin will have these proteins (ATG-13) normalize and this may trend with a reduction in ME/CFS symptoms including PEM flairs. We have published 4 studies building evidence for the role of autophagy disruption in PEM, including results of Phase 1 of this trial.
We are also grateful for the collaboration of the Milwaukee Institute of Drug Discovery (MIDD), University of Wisconsin-Milwaukee (UWM), Indiana Health University ICBI.
FAQs
-
In order to enroll in this trial, you must be a patient under the care of the participating clinicians: Dr. David Kaufman, MD, Dr. Daniel Peterson, MD, Dr. Bela Chedda, MD, Jen Bell, FNP, Braydon Yellman, MD and other participating clinicians. Do you want to add your clinician? Please contact trials@simmaron.com If you are on Rapamycin and have ME/CFS, or you have been told by your doctor that they plan to start you on Rapamycin, please email connect@simmaron.com.
-
18-75 years old
Have a diagnosis of ME/CFS or Long-COVID
Meet criteria for rapamycin therapy (labs & provider approval)
Be willing to provide informed consent to participate in research
-
Longitudinal: 500+
RAPA STEP 4 ME & COVID (double=blind, placebo-controlled) : 40
-
Longitudinal: No, this is a pilot study to observe the effects of low-dose rapamycin treatment, and to determine the incidence of elevated levels of inactive ATG-13 and other autophagy proteins before, during, and after treatment.
RAPA STEP 4 ME&COVID: Yes. Half of the cohort will receive rapamycin for 90 days. The other half will receive a placebo for the first 60 days and then receive rapamycin for the remaining 90 days.
-
· Longitudinal: Minimum study period is 90 days. Maximal observation time is 13 months.
· RAPA STEP 4 ME & PASC (placebo-controlled): 120 days.
-
All results will be published in open access license free peer-reviewed journals
Phase I Longitudinal Observational Study published in JTR
Phase II Longitudinal Observational Study to be published 2Q 2026
RAPA STEP 4 ME & PASC (placebo): Mid 2027; earlier if funded (180 days)